|
Read about electron configurations to learn more. This gives them a full S subshell, which is a pretty stable configuration. For example, metals often lose enough electrons to leave two valence electrons remaining (example: carbon family with +2 charge). It is an icon of chemistry and is widely used in physics and other sciences. Most elements have the ability to form more than one charge, but there are patterns to be found there, as well. The periodic table, also known as the periodic table of the elements, is an ordered arrangement of the chemical elements into rows ('periods') and columns ('groups').You can check out this fancier Periodic Table to see the most common charges of most of the elements.Therefore, sometimes that row is thought of as having “+/- 4” charge, which fits nicely between the +3 and -3 columns. But it’s possible for carbon and silicon to form a C 4- / Si 4- ions, and all of that family’s elements can form +4 ions. The elements in the carbon family aren’t listed as +4 or -4 charged ions, because they are more likely to form covalent bonds instead of being ions.For example, iron can form Fe 2+ and Fe 3+ ions, depending on the situation. 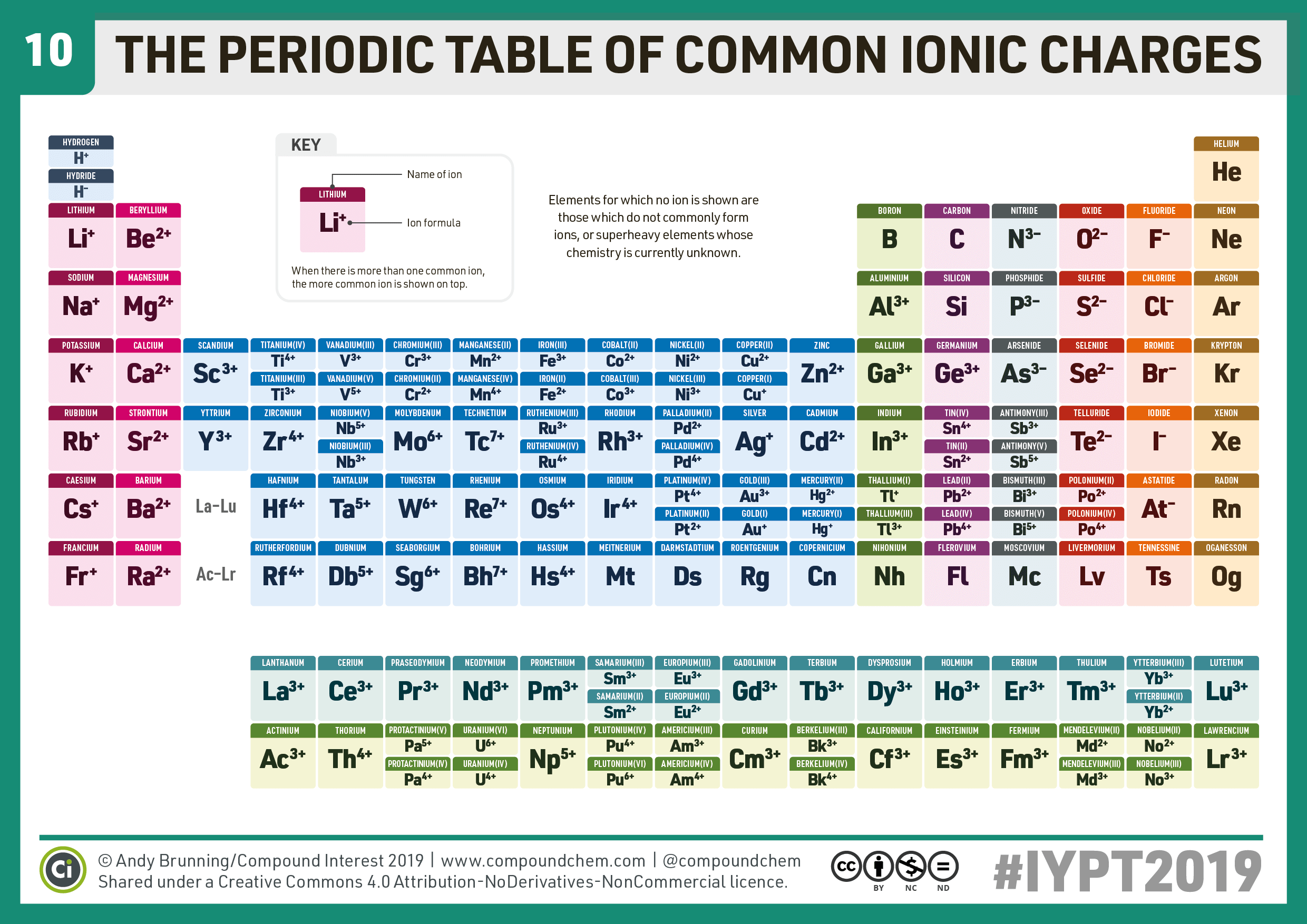
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
